Uses and Applications
Boc-L-Alanine Methyl Ester is widely used in chemical biology research, custom peptide synthesis, and pharmaceutical development.
- Peptide Synthesis: Acts as a key building block (amino acid derivative) that protects the L-alanine moiety during the assembly of chirally-pure peptide sequences.
- Pharmaceutical Intermediates: Utilized in the synthesis of peptidomimetic inhibitors (e.g., cytomegalovirus protease inhibitors) and various bioactive compounds.
- Bioconjugation: Acts as a linker or intermediate in the synthesis of prodrugs and bioconjugates, including Hepatitis C virus inhibitor intermediates.
- Research in Neuroscience: Used in the synthesis of neurotransmitter analogs.
- Material Science: Employed in the creation of specialized polymers and hydrogels for biomedical applications (e.g., drug delivery).
- Stereoselective Reactions: Can serve as a ligand in chemical reactions, displaying stereoselective properties.
Reactivity
The Boc group allows for selective, mild deprotection, making it compatible with various coupling reagents used in organic synthesis. It is relatively stable under normal conditions but should be kept away from strong oxidizing agents.
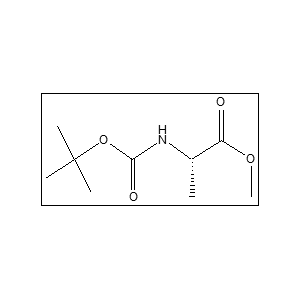
The CAS number is 28875-17-4. The CAS number is 28875-17-4. It is widely used in peptide synthesis, pharmaceutical intermediates, and organic synthesis. The Boc group protects the amino group during chemical reactions, preventing side reactions and enabling selective synthesis. The methyl ester protects the carboxyl group and allows controlled deprotection in later synthesis steps.