Uses and Applications
Boc-L-serine is a versatile building block, particularly in pharmaceutical development and biotechnology:
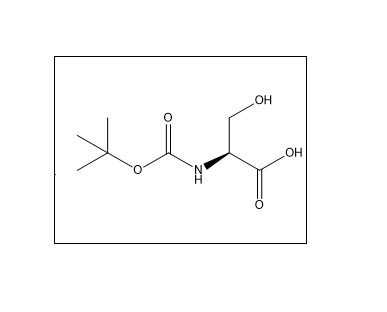
Peptide Synthesis: Used as a protected amino acid in solid-phase peptide synthesis (SPPS) and liquid-phase synthesis to introduce serine residues selectively.
Pharmaceutical Development: Employed in synthesizing bioactive peptides and drugs, particularly those targeting neurological disorders.
Protein Engineering: Used to modify protein structures to improve stability and functionality.
Bioconjugation: Valuable for attaching functional groups to biomolecules.
Synthesis of Intermediates: Acts as a starting material for preparing 𝛼-amino acids and modified serine analogs (e.g., via 𝛽-lactone intermediates).
Biochemical Studies: Used in investigating enzyme mechanisms, especially serine-based enzymes.
Synthesis Examples
It is used as a building block for specialized amino acids and peptide mimics, such as 2-(N-Fmoc)-3-(N-Boc-N-methoxy)-diaminopropanoic acid and Boc-Ser-Leu-OMe.
Safety
It is generally considered a non-hazardous substance according to typical chemical regulations (e.g., Regulation (EC) No. 1272/2008). However, it is classified under “Combustible Solids” and should be handled with standard lab safety equipment (eyes shields, gloves, N95 masks).
Boc-L-Serine is an N-protected amino acid where the amino group of L-serine is protected by a tert-butoxycarbonyl (Boc) group, commonly used in peptide synthesis. The CAS number of Boc-L-Serine is 3262-72-4. It is primarily used in peptide synthesis, pharmaceutical intermediates, and organic chemistry research. The Boc group protects the amino group during chemical reactions, preventing unwanted side reactions and enabling selective synthesis. Yes, it is widely used in both laboratory and large-scale pharmaceutical manufacturing processes. It should be stored in a cool, dry place, away from moisture and direct sunlight, in a tightly sealed container.