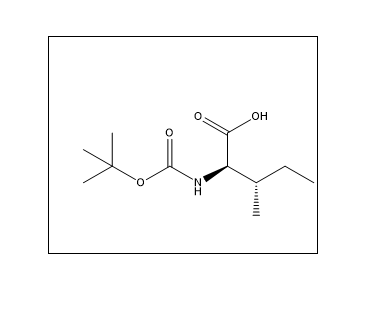
Synonyms
- N-(tert-Butoxycarbonyl)-L-isoleucine
- Boc-L-Ile-OH
- N-Boc-L-isoleucine
- (2S,3S)-2-[(tert-butoxy) carbonyl amino]-3-methylpentanoic acid
- Boc-Ile-OH Hemihydrate
Uses and Applications
Peptide Synthesis: Primary use as a building block in Boc solid-phase peptide synthesis (SPPS) and liquid-phase synthesis. The Boc group is easily removed under mild acidic conditions to reveal the free amino group for chain extension.
Pharmaceutical Research: CASNO: 13139-16-7, Used in the development of new drugs and protease inhibitors, particularly for creating complex active pharmaceutical ingredients (APIs).
Chiral Building Block: Employed as a chiral auxiliary or reagent in asymmetric synthesis to produce enantiomerically pure molecules.
Biotechnology: Used to modify proteins to improve their stability and activity.
Analytical Chemistry: Utilized as a standard or derivative in chromatographic methods to study amino acid metabolism.
Storage
- It is generally recommended to store Boc-L-Isoleucine at 0-8°C (keep cold), although it is stable at room temperature in closed containers.
Boc-L-Isoleucine is a protected amino acid used in peptide synthesis, where the Boc group protects the amine group during reactions. The CAS number is 13139-16-7, which uniquely identifies this compound. It is widely used in peptide synthesis, pharmaceutical research, and the development of active pharmaceutical ingredients (APIs). It should be stored in a cool, dry place in tightly sealed containers away from moisture and direct sunlight. Yes, it is commonly used in laboratory research and industrial R&D processes.