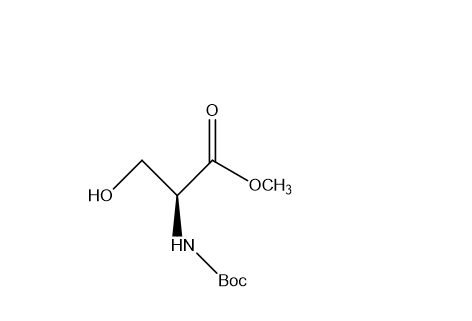
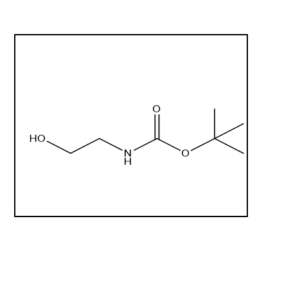
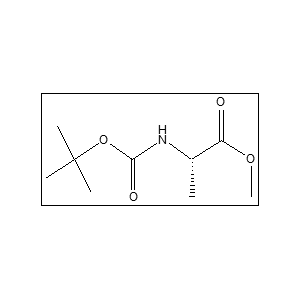
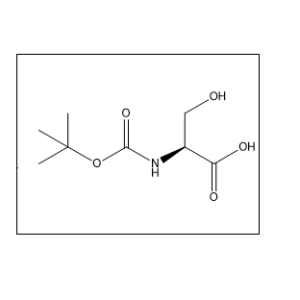
Uses:
- Organic Synthesis: It is used to synthesize important organic reagents, such as
(2-methylthio-1–toluenesulfonyl)methanesulfonamide and tutyl ((2-(trimethylsilyl) ethyl)sulfonyl)carbamate.
- Nitrogen Source: It acts as a nitrogen source for converting carboxylic acids into their corresponding nitriles.
- Catalysis: In Sharpless dihydroxylation, it functions as a catalyst during the final hydrolysis step to help form cis-vicinal-diols.
- Solvent: It is sometimes employed as a solvent in specific dihydroxylation reactions
Key Applications:
- Chemical Synthesis:Acts as an intermediate for creating various compounds, including indole-N-acetamide derivatives and other pharmaceuticals.
- Pharmaceutical Research:Used in the development of drug candidates, such as pyrimidine-substituted 3,5-dihydroxy-6-heptenoates and the anti-inflammatory drug repertaxin.
- Reagent in Laboratory Studies:Used in biochemical research to study protein-ligand complex binding affinities.
- Materials Synthesis:Utilized in reactions involving thionyl chloride, cyanates, carbon disulfide, ketones, and aldehydes.
It is mainly used in peptide synthesis, pharmaceutical research, and organic chemistry applications. The CAS number is 3144-09-0 Yes, it is widely used in research laboratories for peptide and drug development studies.