Uses and Applications
- Peptide Synthesis: Serves as a protected, activated amino acid intermediate in solid-phase and solution-phase synthesis for creating Proline-containing peptides.
- Pharmaceutical Development: Key intermediate in synthesizing peptidomimetics and bioactive molecules, particularly in designing enzyme inhibitors.
- Chemical Research: Utilized in studies requiring chiral, N-protected amino acid derivatives.
- Bioconjugation: Used as a building block for specialized, tailored amino acid structures in advanced biological research.
Safety and Handling
- Classification: No hazardous classification required under typical US OSHA regulations.
- Storage: Generally stored at room temperature or 2–8°C; stable under recommended conditions.
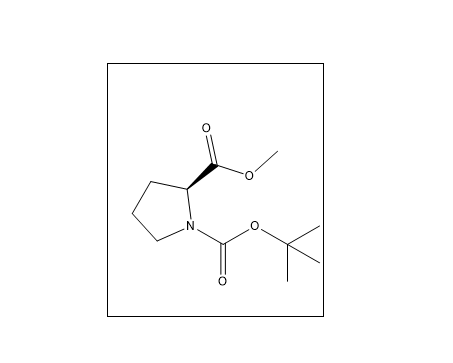
Boc-L-Proline Methyl Ester is an N-protected derivative of L-proline, where the amino group is protected by a tert-butoxycarbonyl (Boc) group and the carboxyl group is esterified. Boc-L-Proline Methyl Ester is an N-protected derivative of L-proline, where the amino group is protected by a tert-butoxycarbonyl (Boc) group and the carboxyl group is esterified. The CAS number is 59936-29-7. It is widely used in peptide synthesis, pharmaceutical intermediates, and organic chemistry research. The Boc group protects the amino group during synthesis, preventing unwanted reactions and enabling stepwise peptide formation. The methyl ester protects the carboxyl group and can be removed when needed during later stages of synthesis.