Uses and Applications
Wittig Reaction (Olefination): It is primarily used to react with aldehydes to form substituted methyl acrylates, allowing for two-carbon homologation.
Synthesis of Pyrazoles: It is utilized in the efficient synthesis of pyrazoles via reaction with methyl diazo acetate in the presence of triethyl amine.
Preparation of Further Reagents: It is used in the preparation of (triphenyl phosphoranylidene) ketene.
Pharmaceutical/Chemical Research: It acts as a versatile intermediate for constructing complex molecular architectures, particularly in the development of agrochemicals and pharmaceuticals.
Analytical and Material Science: It is used as a reagent in studies involving phosphorus chemistry, catalyzing new reactions, and designing specific functional materials.
Handling and Storage
Storage: Store in a cool, dry place, typically under inert gas (nitrogen) to avoid air sensitivity.
Storage Temperature: Room temperature, but recommended in a cool, dark place
Appearance: White to pale cream powder.
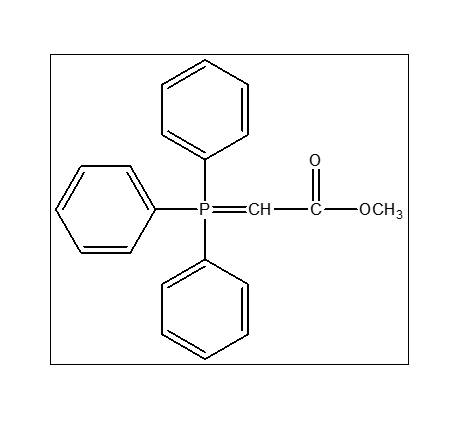
Methyl (triphenyl phosphoranylidene)acetate
This reagent is known for its high stability and ease of handling compared to other Wittig reagents, making it ideal for both laboratory and industrial applications.
Methyl (triphenyl phosphoranylidene) acetate (CAS 2605-67-6) is a stable organo phosphorus compound primarily utilized as a Wittig reagent in organic synthesis to convert aldehydes and ketones into
𝛼,-unsaturated methyl esters (two-carbon homologation). It typically appears as a white to off-white crystalline powder.
It is mainly used as a Wittig reagent to form carbon–carbon double bonds in organic synthesis. The CAS number is 2605-67-6. Yes, it is widely used in organic chemistry research and synthesis. Store in a cool, dry place, tightly sealed, and protected from moisture and air. It typically appears as a white to off-white crystalline solid.