Safety and Handling
- Hazard Class: Class 3 (Flammable liquid)
- Storage: Keep in a cool, dark place, ideally below 15°C.
- Hazard Statements: H226 (Flammable liquid and vapour), H315 (Causes skin irritation), H319 (Causes serious eye irritation), H335 (May cause respiratory irritation).
- Precautionary Statements: P210 (Keep away from heat), P280 (Wear protective gloves/clothing/eye protection).
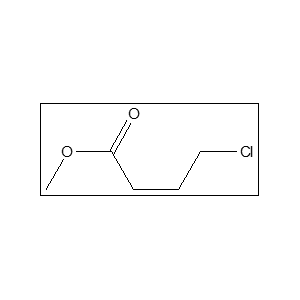
Methyl 4-chlorobutyrate (CAS 3153-37-5) is a colorless, combustible liquid (𝐶5𝐻9𝐶𝑙𝑂2) mainly used in pharmaceuticals and synthesis. Key synonyms include Methyl 4-chlorobutanoate, 4-Chlorobutyric acid methyl ester, and Methyl 𝛾-chlorobutyrate. It has a boiling point of 175-176∘C and acts as an important intermediate for chemical production.
Synonyms and Identifiers
- Methyl 4-chlorobutanoate
- 4-Chlorobutyric acid methyl ester
- Methyl 𝛾-chlorobutyrate (or Methyl gamma-chlorobutyrate)
- Methyl omega-chlorobutyrate
- Butanoic acid, 4-chloro-, methyl ester
- ClCH22(CH22)$_2𝐶(𝑂)𝑂𝐶𝐻_3$
- CAS Number: 3153-37-5
- EC Number: 221-592-9
- MDL Number: MFCD00001003
- PubChem CID: 76612
Methyl 4-chlorobutyrate (CAS 3153-37-5) is a versatile, colorless chemical intermediate primarily used to synthesize pharmaceuticals (such as Prasugrel for blood clotting), agrochemicals, and fine chemicals. It acts as a building block for complex organic molecules, allowing the introduction of functional groups via nucleophilic substitution.
Key Uses and Applications:
- Pharmaceutical Intermediate:Used in the production of Active Pharmaceutical Ingredients (APIs) for cardiovascular treatment, antiviral, antibacterial, and antifungal medications.
- Chemical Synthesis:Acts as a key building block in organic chemistry due to its reactivity in nucleophilic substitution, ester hydrolysis, and Grignard reactions.
- Agrochemicals:Utilized in the development and manufacturing of agricultural chemicals.
- Specialty Chemical Manufacturing:Employed in producing fine chemicals and potentially in fragrance applications.
- Laboratory Reagent:Used in research for constructing carbon chains and new compound development.