Featured Headlines
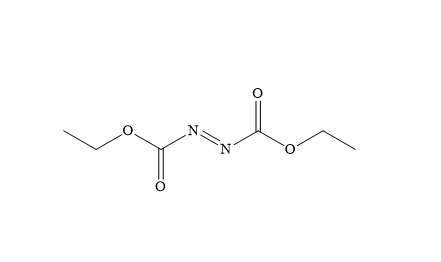
Di ethyl Azodicarboxylate (DEAD) CASNO: (1972-28-7)
Physical properties:
Molecular Formula: C6H10N2O4
Appearance: Orange-red liquid
Density: 1.106 g/mL at 25°C
Boiling Point: 106°Cat 13 mmHg
Melting Point: 6°C
Refractive Index: 1.47 n20/D
Flash Point: 85°C
Stability: Stable but light-sensitive and potentially explosive if heated under confinement
Solubility: Soluble in many organic solvents
Synonyms:
- DEAD (Diethyl Azodicarboxylate)
- Diethyl diazenedicarboxylate
- Diethyl azodiformate
- Azodicarboxylic acid diethyl ester
- 1,2-Diazenedicarboxylic acid, 1,2-diethyl ester
- 1,2-Ethoxycarbonyl diazene
- Diethoxycarbonyldiazene
- Ethyl (NE)-N-ethoxycarbonyliminocarbamate
- DEADCAT
Uses:
- Mitsunobu Reaction: Extensively used to convert alcohols into various functional groups (amines, azides, esters, thioethers).
- Organic Synthesis: Acts as a reagent in the alpha-thiocyanation of enolizable ketones and annulation of N-protected imines.
- Diels-Alder Reactions: Used as a strong dienophile.
- Pharmaceutical Synthesis: Used in the preparation of specialized drugs, including zidovudine (AIDS drug) and antitumor agents.
- Dehydrogenating Agent: Used to convert alcohols to carbonyl derivatives and hydrazines.
- Material Science: Employed in polymer chemistry for modifying polymer properties through functionalization.
Key Applications:
- Mitsunobu Reaction:Acting as a core reagent for synthesizing esters, ethers, thioethers, and amines from alcohols.
- Pharmaceutical Synthesis:Used in the production of complex APIs and intermediates, such as developing antiviral or antitumor agents.
- Organic Transformations:Utilized in Diels-Alder reactions and as a dehydrogenating agent (e.g., in converting alcohols to carbonyls).
- Material Science:Employed as a radical polymerization initiator and in the preparation of fluorescent materials.
- Biochemical Research:Used in the preparation of cellotriose analogues and cannabinoid CB1 receptor ligands.