Structure:
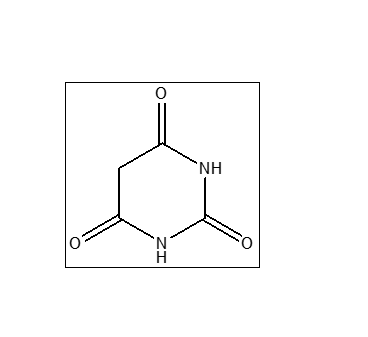
Barbituric acid (CAS 67-52-7), Also known as 2,4,6-trihydroxypyrimidine or malonylurea, is an organic compound based on a pyrimidine heterocyclic skeleton. It is widely used as a synthetic intermediate in the pharmaceutical, plastic, and chemical industries. Although it is the parent compound of barbiturate drugs, unsubstituted barbituric acid itself possesses no central nervous system (CNS) sedative or hypnotic properties.
Properties
CAS Number: 67-52-7
Molecular Formula: C4H4N2O3
Molecular Weight: 128.09 g/mol
Appearance: White to off-white, light beige powder or crystals
Melting Point: 248°C to 255°C (with decomposition)
Solubility: Soluble in water (142 g/L at 20°C) and water/methanol mixtures; slightly soluble in alcohol and ether
Acidity: Strong acid in aqueous medium (pKa 4.01)
Reactivity: Active methylene group allows for Knoevenagel condensation reactions
Synonyms
2,4,6(1H,3H,5H)-Pyrimidinetrione
Malonylurea
6-Hydroxyuracil
2,4,6-Trioxohexahydropyrimidine
Pyrimidine-2,4,6-triol
1,3-Diazinane-2,4,6-trione
Uses and Applications
Barbituric acid serves primarily as a versatile structural block in organic synthesis, rather than as an end-product itself.
Pharmaceutical Industry:Precursor for Barbiturates: Used to synthesize sedative, hypnotic, anesthetic, and anticonvulsant agents, such as phenobarbital, pentobarbital, and thiopental.
Vitamin B2 Synthesis: Acts as an active ingredient in the production of Riboflavin (Vitamin B2).
Drug Intermediate: Used in the preparation of anticancer agents, antiviral drugs, and anti-inflammatory compounds.
Chemical and Industrial Synthesis:
Knoevenagel Condensation: Used to produce 5-arylidene barbituric acids through reactions with aromatic aldehydes.
Manufacturing: Utilized in the creation of plastics, textiles, and polymers.
Analytical Chemistry:
Electrochemistry: Employed in the electrochemical oxidation of iodine, using techniques like cyclic voltammetry and controlled potential coulometry.
Colorimetric Analysis: Used for the detection of furfural derivatives.
Chemical Structure Note
Barbituric acid exists primarily in its trioxo form (2,4,6 (1H,3H,5H)-pyrimidinetrione) but is often referred to by its tautomeric forms, such as 2,4,6-trihydroxypyrimidine.