Uses and Applications
Wittig Reagent: It is widely used in chemical synthesis to convert aldehydes and ketones into ethyl 2-methyl-2-alkenoates.
Pharmaceutical Intermediates: Used in the production of API (Active Pharmaceutical Ingredients) and antiviral compounds.
Agrochemicals: Serves as a building block in the development of agricultural chemical compounds.
Synthetic Organic Chemistry: Functions as a tool for forming C-C bonds in complex synthesis, such as in the production of natural products (e.g., malevamide E derivatives).
Material Science: Used in research involving the synthesis of specialized polymers and coatings.
Safety and Handling
Signal Word: Warning
Hazard Statements:
H302: Harmful if swallowed
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
Precautions: Air-sensitive; requires storage under inert gas (e.g., nitrogen or argon) in a freezer.
Production
Ethyl 2-(triphenylphosphoranylidene) propionate is typically prepared by the reaction of ethyl 2-bromopropanoate with triphenylphosphine, followed by treatment with a base (such as sodium hydroxide) to form the phosphorane.
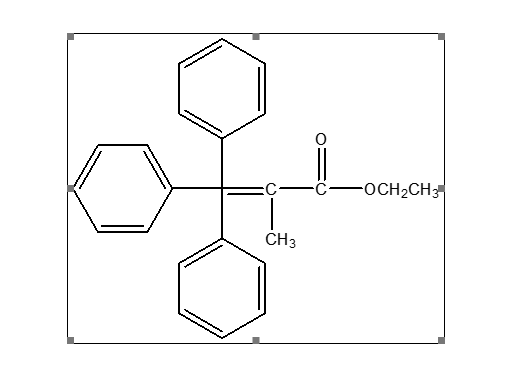
Ethyl 2-(triphenylphosphoranylidene) propionate (CAS 5717-37-3) is a stabilized Wittig reagent primarily used in organic synthesis for the conversion of aldehydes and ketones into 𝛼,-unsaturated esters. It is a yellow crystalline powder known for facilitating Carbon-Carbon (C-C) bond formation in the synthesis of pharmaceuticals, agrochemicals, and complex organic molecules.
It is mainly used as a Wittig reagent in organic synthesis to form carbon–carbon double bonds (olefination reactions). The CAS number is 5717-37-3. Yes, it is widely used in organic chemistry research and synthesis.