Uses:
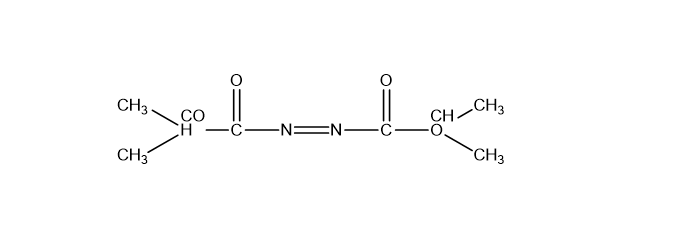
Mitsunobu Reactions: Acts as an azodicarboxylic acid derivative, usually in combination with triphenylphosphine, to facilitate conversions in organic synthesis.
Pharmaceutical Intermediate Production: Used in the creation of various compounds, including potential HIV drugs, anticancer agents, and inhibitors for hepatitis C virus.
Chemical Synthesis: Serves as a reactant in the preparation of chromenes (resembling cannabinoids), norbornene-based polymers, and organic dyes.
Functional Group Manipulation: Acts as a selective deprotecting agent for -benzyl groups.
Radical Trapping: Used in metal-free oxyaminations of alkenes.
Key Applications:
- The Mitsunobu Reaction
DIAD is a crucial component in the Mitsunobu reaction, where it is used alongside triphenylphosphine to convert alcohols into various functional groups (ethers, esters, amines, azides) with high efficiency.
- Pharmaceutical Synthesis and Drug Development
- Active Pharmaceutical Ingredient (API) Intermediate: DIAD is used in the production of various, complex pharmaceutical compounds.
- Specific Drug Candidates: It is used as a reactant in the preparation of:
- MK-3281: A hepatitis C virus NS5B polymerase inhibitor.
- Procarbazine: A chemotherapy medication.
- Zidovudine (AZT): An HIV/AIDS medication.
- Analogs of Quorum-Sensing Molecules: Used in studying immunosuppressive properties.
- Autoimmune & Eye Disorder Research: It is used in the development of treatments for these conditions.
- Material Science and Specialized Chemistry
- Polymer Chemistry: Used to create norbornene-based, guanidine-rich polymers that mimic cell-penetrating peptides (CPPs).
- Organic Dyes: Used in the synthesis of donor-acceptor-donor organic dyes for dye-sensitized solar cells.
- Chemical Synthesis: Acts as a mild, selective oxidizer, particularly useful for removing
n-benzyl protecting groups.