Uses
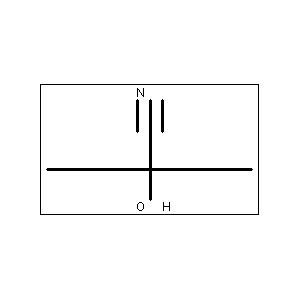
Acetone Cyanohydrin (CAS No: 75-86-5) is primarily used as a key intermediate in the production of methyl methacrylate (MMA), which is essential for manufacturing acrylic plastics and resins. It also acts as a source of hydrogen cyanide (HCN) in various chemical synthesis processes. Additionally, it is used as a raw material in the preparation of insecticides and in specialized organic reactions such as the Strecker reaction.
Key Applications
Cyanation reactions of aryl halides, aldehydes, and alcohols
Stereospecific epoxide ring opening reactions
Strecker reaction of α-amido sulfones to synthesize α-amino nitriles
Intermediate in the manufacturing of acrylic plastics and specialty chemicals
Chemical synthesis processes in pharmaceutical and industrial chemistry