Key Synonyms for CAS 2449-05-0
DBAD
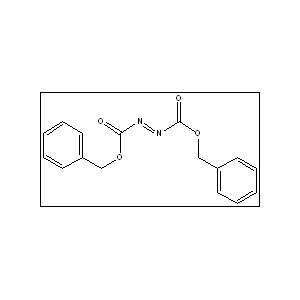
Dibenzyl diazene-1,2-dicarboxylate
Azodicarboxylic acid dibenzyl ester
Bis(benzyloxycarbonyl)diazene
Azodiformic acid dibenzyl ester
Dibenzyl (E)-1,2-diazenedicarboxylate
Benzyl N-(phenylmethoxycarbonyliminocarbamate)
Dibenzyl azodicarboxylate (DBAD, CAS 2449-05-0) is an orange crystalline compound used primarily as a versatile electrophilic reagent in organic synthesis, particularly for Mitsunobu reactions, 𝐶-𝑁 bond formation, and amino acid synthesis. It acts as a coupling agent for pharmaceuticals and in polymer chemistry to produce advanced materials.
Key Uses and Applications
- Organic Synthesis Reagent: DBAD is used for the stereoselective synthesis of amino acids and heterocyclic compounds.
- Mitsunobu Reactions: It is employed as a coupling reagent to facilitate the conversion of alcohols into various functional groups.
- Pharmaceutical Development: It is used in the synthesis of labeled compounds, such as Minocycline-d6
- Polymer and Material Science: It functions as a reagent for creating specialized polymers with high-performance thermal and mechanical properties.
- Photoinitiator: It is used in UV-curable inks and coatings.
Key Characteristics
- Reactivity: It acts as an electrophilic reagent in the synthesis of 𝐶-glycosyl 𝛼-amino acids and for the amination of carbonyl compounds.
- Safety and Storage: It is typically stored at 0-8°C.
The compound is valued for being more stable and easier to handle compared to other azodicarboxylates, making it a preferred choice in complex molecule construction.
Dibenzyl Azodicarboxylate is an azo-based organic reagent commonly used in chemical synthesis, particularly in Mitsunobu reactions. It is used in pharmaceutical intermediates, peptide synthesis, and organic transformations such as esterification and etherification. Yes, it is widely used in both laboratory and large-scale industrial synthesis due to its high efficiency and reliability. Store in a tightly sealed container in a cool, dry, and well-ventilated area, away from heat and direct sunlight.